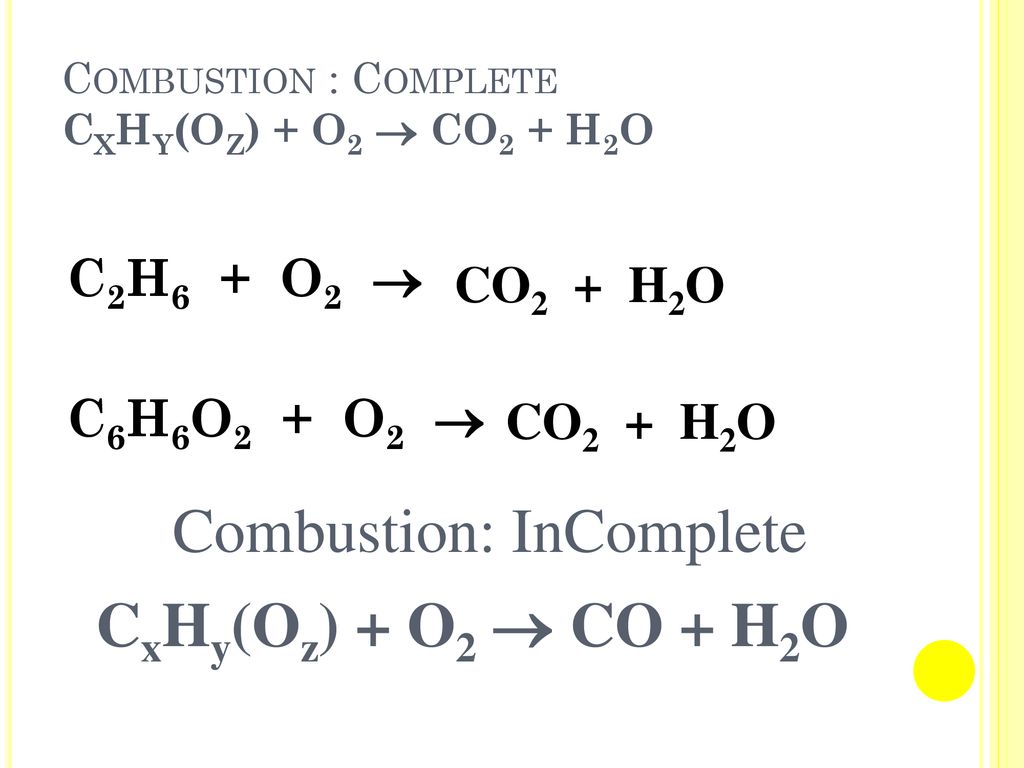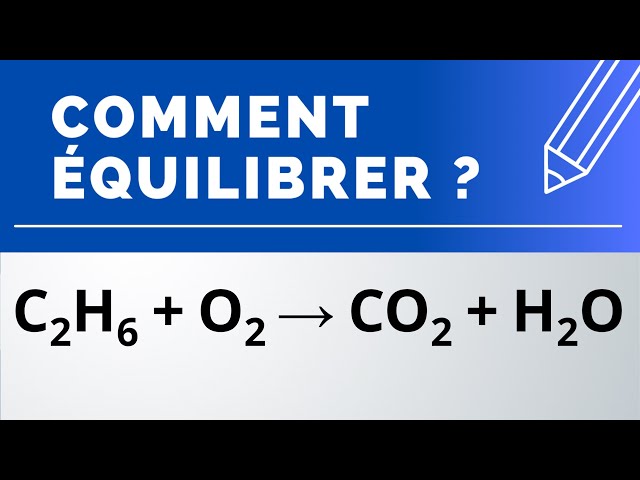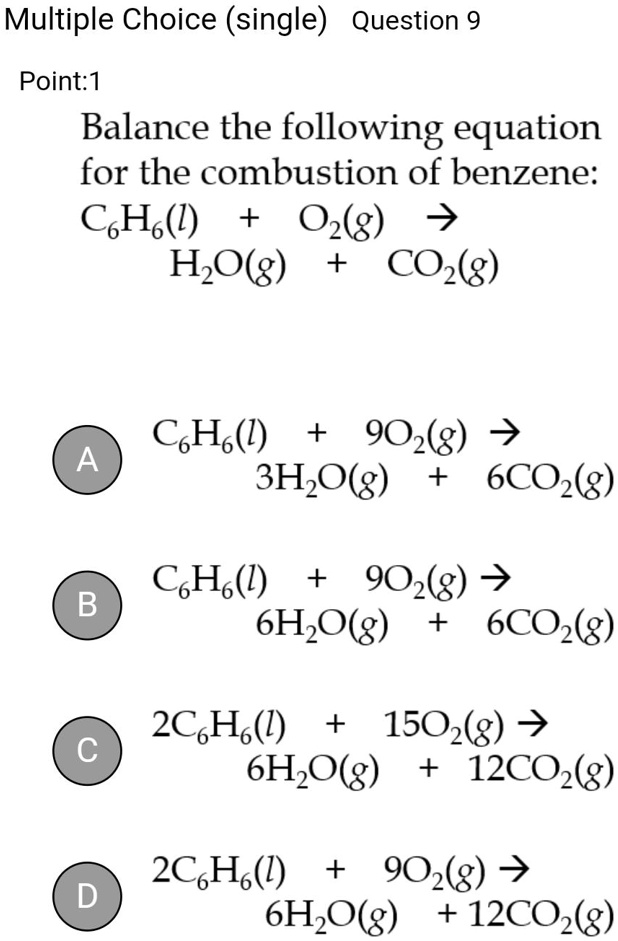
SOLVED: Multiple Choice (single) Question 9 Point: 1 Balance the following equation for the combustion of benzene: C6H6(l) + O2(g) â†' H2O(g) + CO2(g) C6H6(l) + 15O2(g) â†' 6H2O(g) + 6CO2(g) A

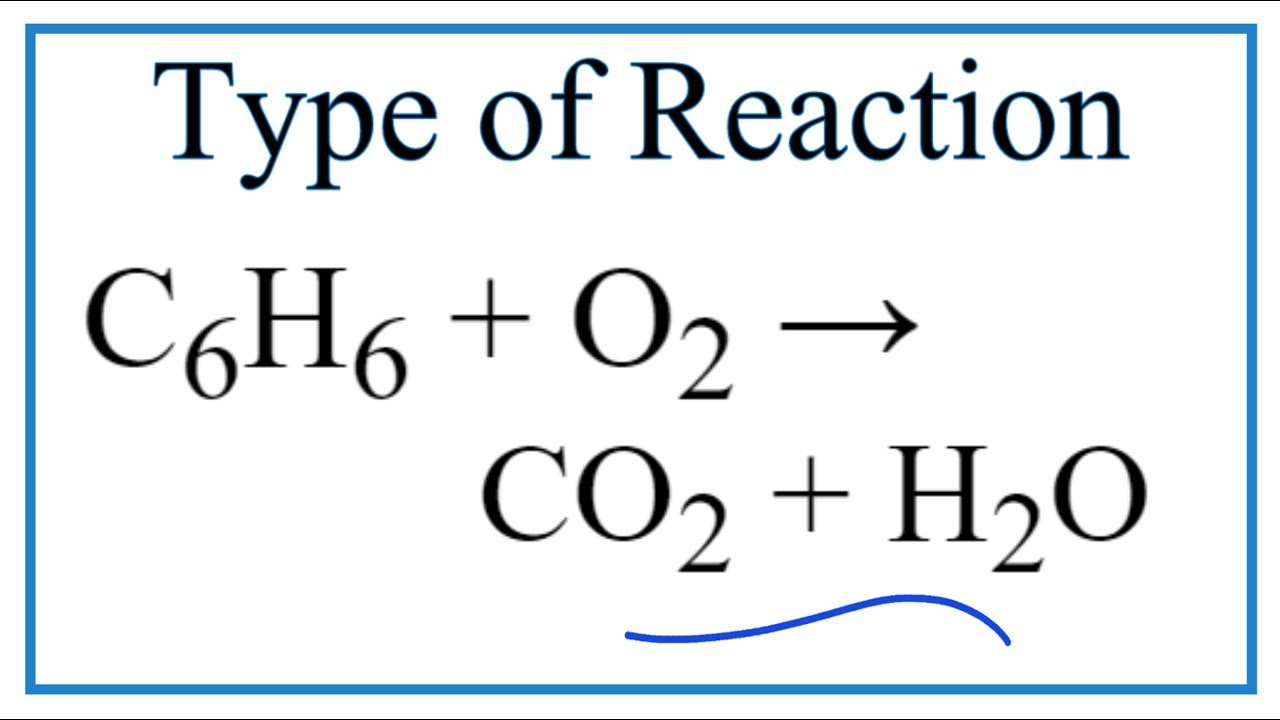
C6H6 +O2 ------}CO2 + H2O balance By oxi Methods - Chemistry - Redox Reactions - 13233357 | Meritnation.com

PPT - Chemical Equations reactants products reactant - starting substance in a chemical reaction PowerPoint Presentation - ID:5812787

SOLVED: If 56.3 g of benzene (C6H6) are combusted with excess oxygen, what mass of carbon dioxide will be obtained? C6H6+O2⋯CO2+H2O
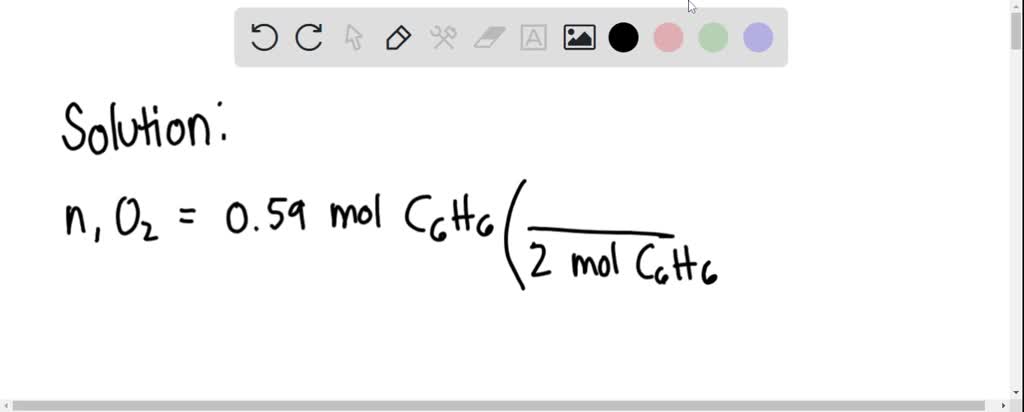
SOLVED: Benzene (C6H6) burns in air according to the following equation shown below. How many moles of O2 are required to react with 0.59 mol of C6H6? 2 C6H6(l) + 15 O2(g)
![Which set of conditions are correct for the reaction below? 2C_3H_8+7O_2to 6CO+8H_2O A. co [algebra] Which set of conditions are correct for the reaction below? 2C_3H_8+7O_2to 6CO+8H_2O A. co [algebra]](https://p16-ehi-va.gauthmath.com/tos-maliva-i-ejcjvp0zxf-us/416b1902cada4902a7141d7b6e990ae8~tplv-ejcjvp0zxf-webp.webp)
Which set of conditions are correct for the reaction below? 2C_3H_8+7O_2to 6CO+8H_2O A. co [algebra]

Drill #4B 2/10/15 Predict the products and balance the equation for the following double-replacement reaction: Na2CrO4 (aq) + Ba(OH)2 (aq) - ppt download